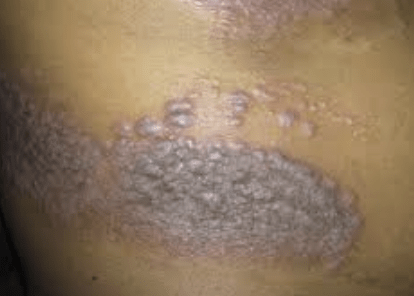
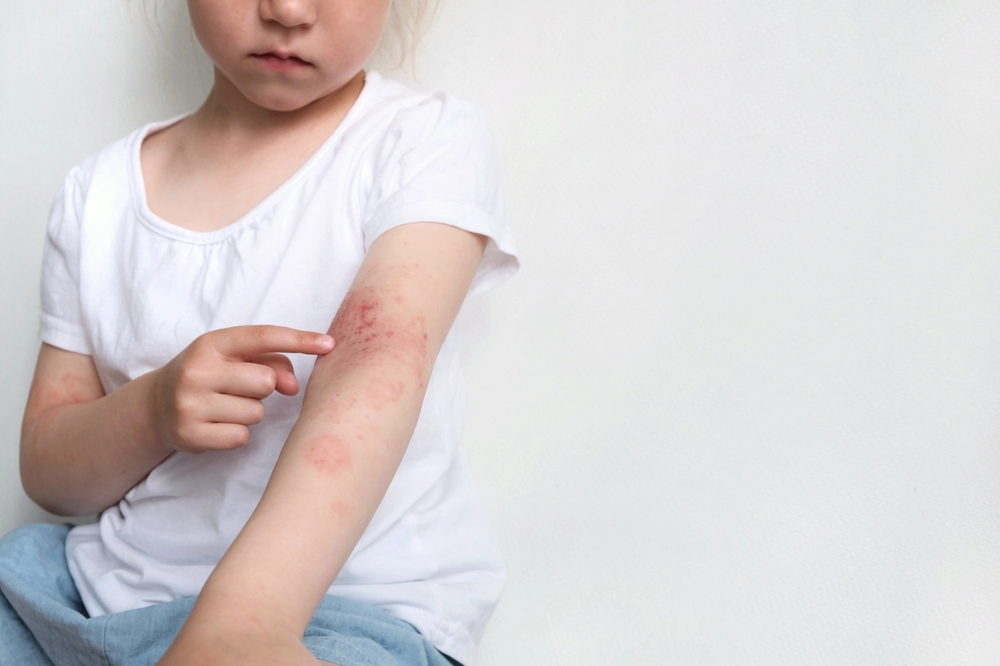
In a study, the drug ixekizumab, an immunoglobulin G subclass 4 monoclonal antibody, was found highly effective and safe for up to five years in treating patients with moderate-to-severe psoriasis or psoriatic arthritis (PsA). The researchers, who followed participants in a real-life cohort, aimed to evaluate the drug’s efficacy, survival, and safety over this extended period. The study was conducted retrospectively at the Dermatologic Clinic of the University of Turin, assessing participants who received at least one dose of ixekizumab between January 2017 and March 2021, with follow-up until April 2023. The team examined the drug’s effectiveness at several intervals, specifically at 16, 52, 104, 156, 208, and 260 weeks, using PASI scores and other measures.
Findings from the study showed by the 260-week mark, there was a 65.5% drug survival rate among patients, with those achieving a rapid response having a lower risk of treatment discontinuation. The results indicated 54.1% reached PASI100, 60.5% reached PASI90, and 73% reached PASI ≤3 by the 16-week mark, improving to 59.1%, 81.8%, and 95.5%, respectively, by the end of the study. A higher mean body mass index was significantly associated with a decreased likelihood of achieving these outcomes by earlier time points. The researchers concluded that ixekizumab showed high effectiveness and safety for up to five years, with a rapid response to treatment being predictive of long-term response.
Reference: Smith T. Ixekizumab Shown to be Safe, Effective for Up to 5 Years for Psoriasis. HCPLive. Published January 2, 2024. Accessed March 13, 2024. https://www.hcplive.com/view/ixekizumab-shown-to-be-safe-effective-for-up-to-5-years-for-psoriasis
Link: https://www.hcplive.com/view/ixekizumab-shown-to-be-safe-effective-for-up-to-5-years-for-psoriasis